Paradigm shift in cancer therapy: targeting non-dividing cancer cells to prevent cancer recurrence.
We develop monoclonal antibodies and antibody-drug conjugates (ADCs) to disrupt the metabolic reactivation of non-dividing dormant cancer cells to prevent cancer recurrence.
Science behind our technology
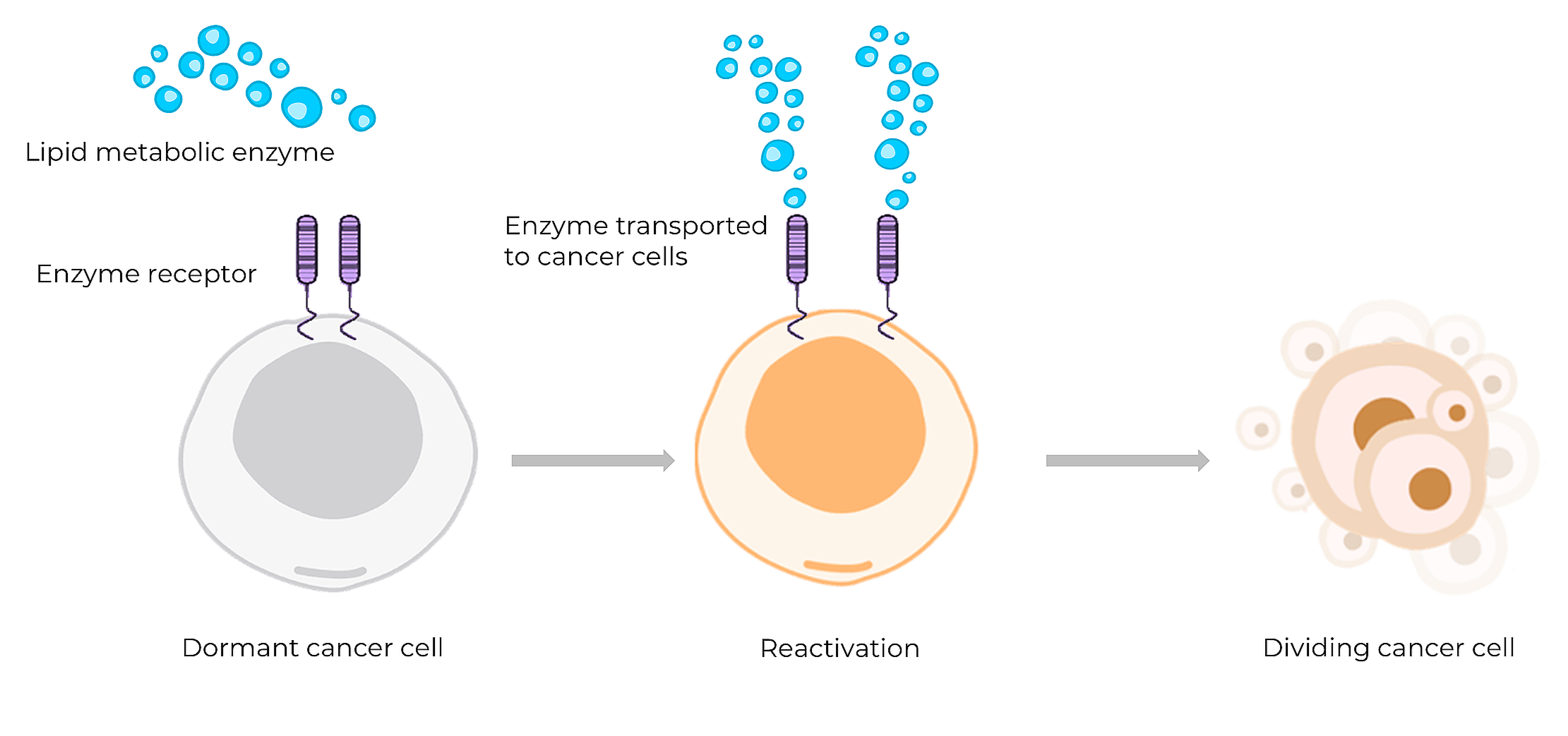
We are focused on preventing cancer recurrence by eliminating dormant (non-dividing) cancer cells which cause cancer relapse. Dormant cancer cells persist in tumor microenvironment (TME) as minimal residual disease, escaping initial therapies which target proliferative cells. The cancer cell dormancy is sustained through dynamic interactions within TME, where dormant cells rely on microenvironment-derived survival mechanisms and metabolic adaptations. In particular, dormant cancer cells exhibit a strong dependence on lipid metabolism for long term survival. Reactivation of dormant cancer cells, which is an essential step to lead to cancer recurrence, is initiated by lipid metabolism activation events.
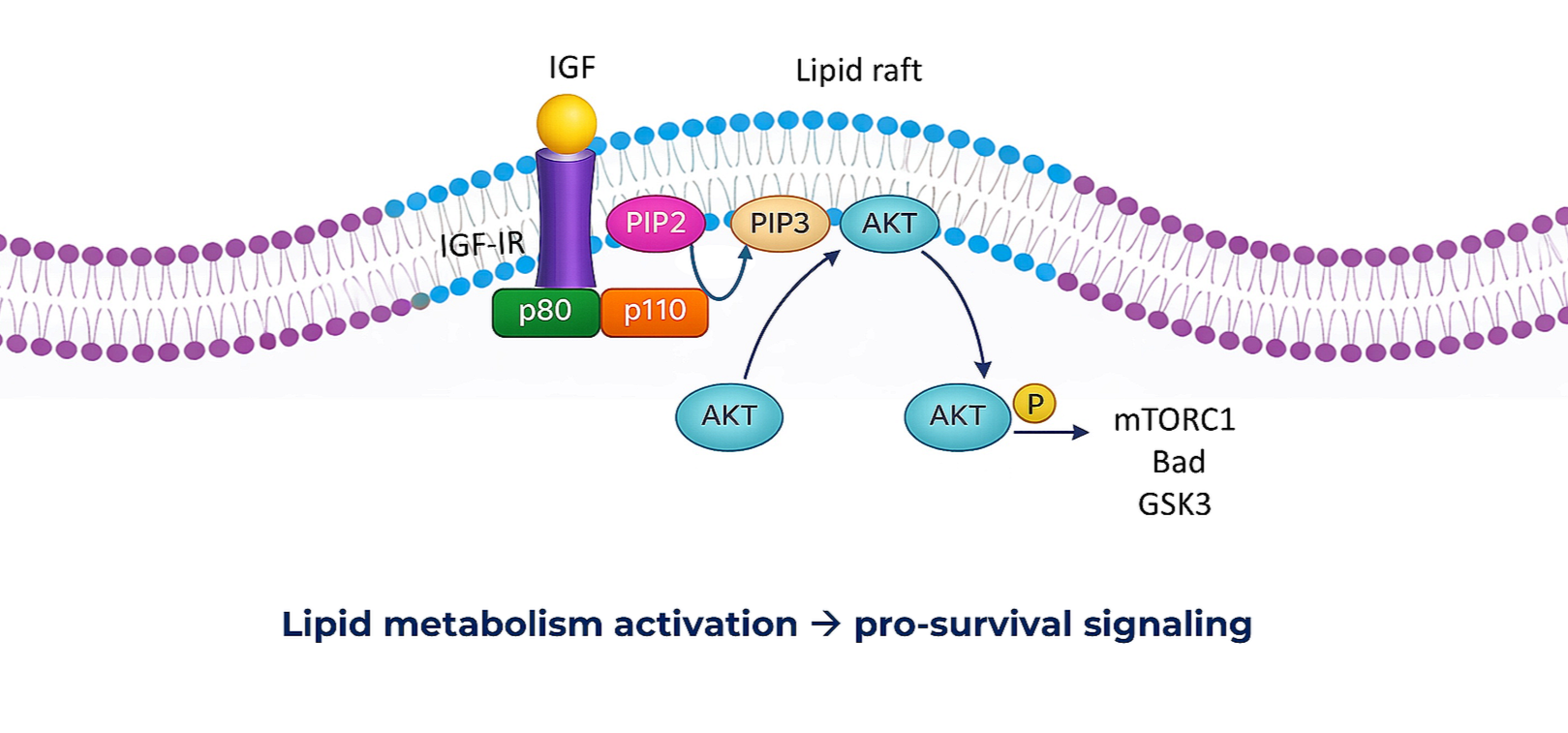
We exploit metabolic dependence of dormant cancer cells within TME, by targeting tumor-specific antigens and proteins that help the survival and reactivation of dormant cancer cells. We identified a pair of targets- a metabolic enzyme and its receptor which are essential for dormant cancer cell reactivation. The enzyme transport through the receptor enables lipid metabolism, leading to formation of lipid rafts and activation of pro-survival signaling platforms.
By disrupting these metabolic pathways, we aim to prevent the transition from dormant to dividing cancer cells, thereby preventing cancer relapse and improving long-term patient outcomes.
Therapeutic Platforms
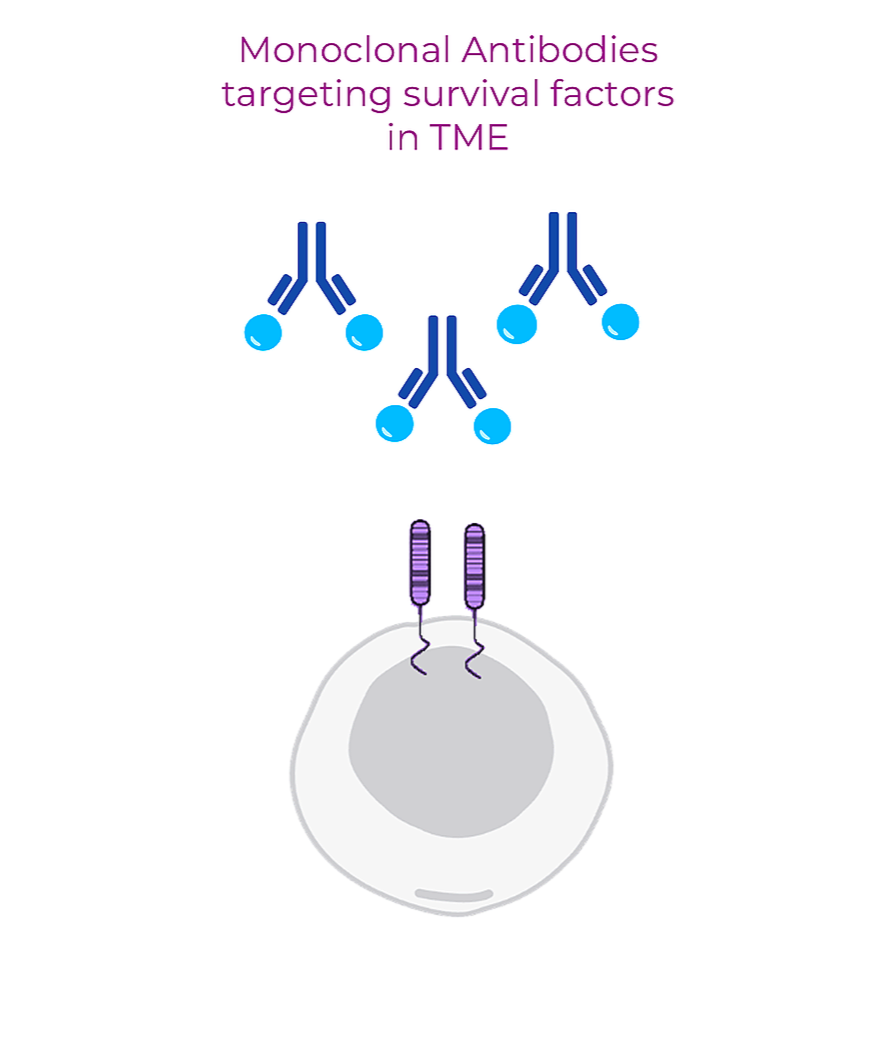
Neutralizing Antibody Platform
We develop first-in-class monoclonal antibodies against essential survival factors in the tumor microenvironment (TME). These antibodies are engineered to neutralize cancer-specific secreted factors and block the cancer cell transition from dormant to active state.
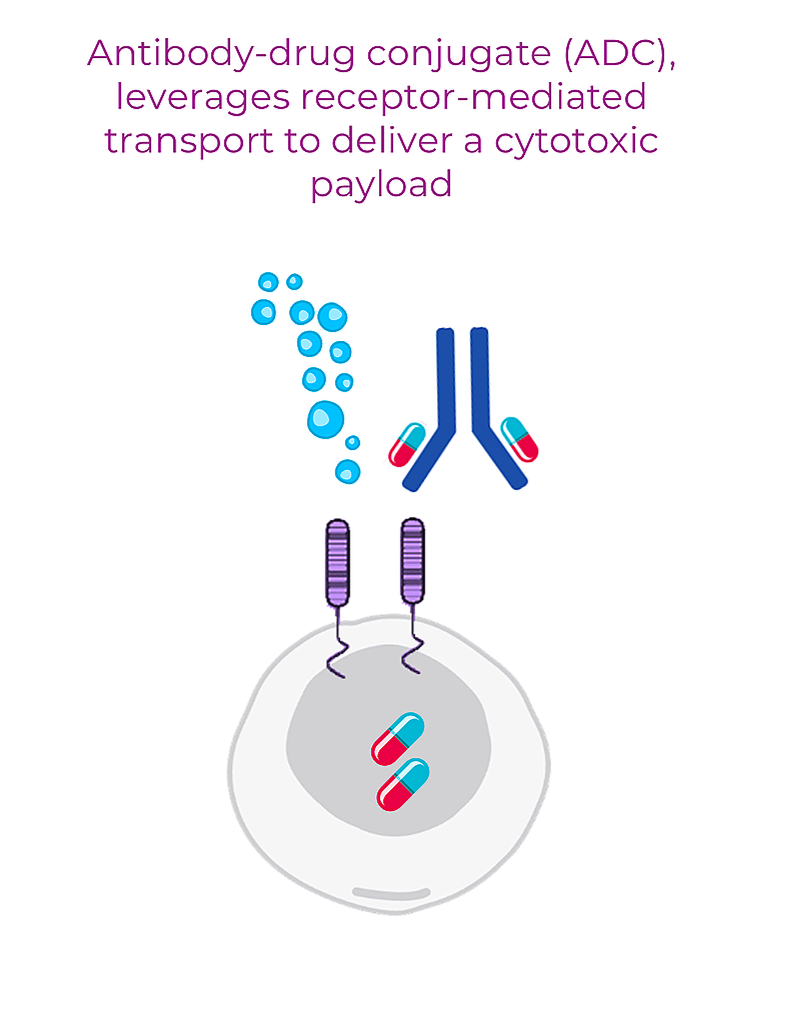
Receptor Targeted Drug Delivery-ADC Platform
We develop novel antibodies targeting receptors expressed on the surface of dormant cancer cells. These antibodies can be conjugated to chemical drugs or radiolabeled with diagnostic or therapeutic isotopes to generate antibody-drug conjugate (ADC). ADC is delivered to cancer cells by leveraging receptor mediated transport.
FREZENT is interested in developing ADCs that combine proprietary antibodies with cytotoxic payload directed at blocking dormant cancer cell metabolism. This approach enables cancer-selective metabolism- targeted therapeutic effects.
Pipeline
Indication
Asset
in-vitro PoC
in-vivo PoC
Lead Nomination
Lead Optimization
IND Enabling
Target Validation
FZ07- ADC
HNSCC*
Osteosarcoma
*Head and neck squamous cell carcinoma
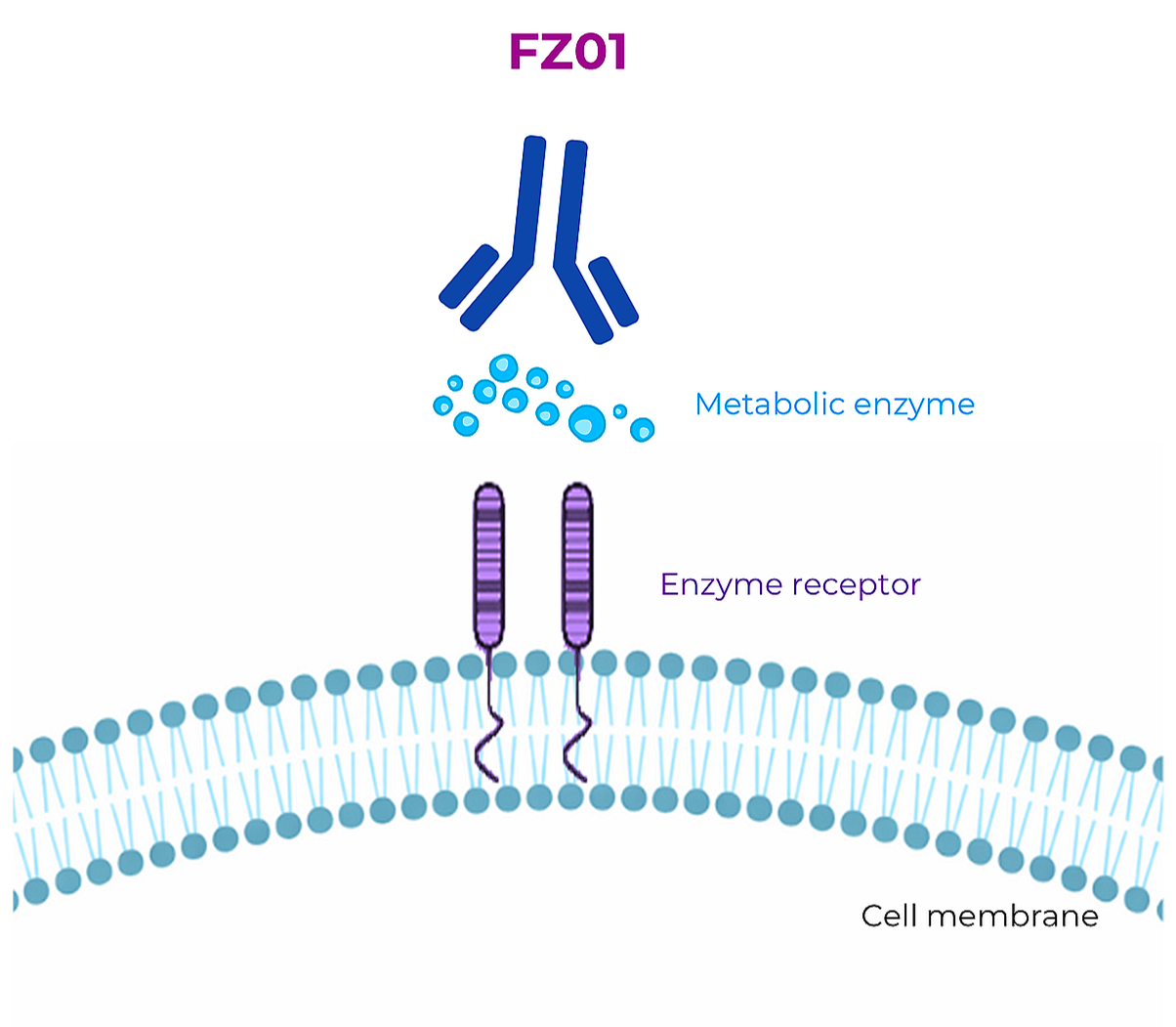
FZ01 - mAb
FZ01
FZ01 monoclonal antibody (mAb) targets a secreted lysosomal enzyme present in the tumor microenvironment. The enzyme is a validated cancer target involved in lipid metabolism and essential for dormant cancer cell reactivation. FZ01 is the first-in-class biological metabolic inhibitor.
FZ01 is in development for clinical use as an adjuvant therapy in combination with standard-of-care chemotherapy or targeted therapy to extend time in remission.
Supporting in-vivo evidence:
FZ01 prevents tumor re-growth after chemotherapy.
FZ01 enhances the effect of cisplatin chemotherapy on tumor growth.
FZ01 is effective against both dividing and non-dividing cancer cells.
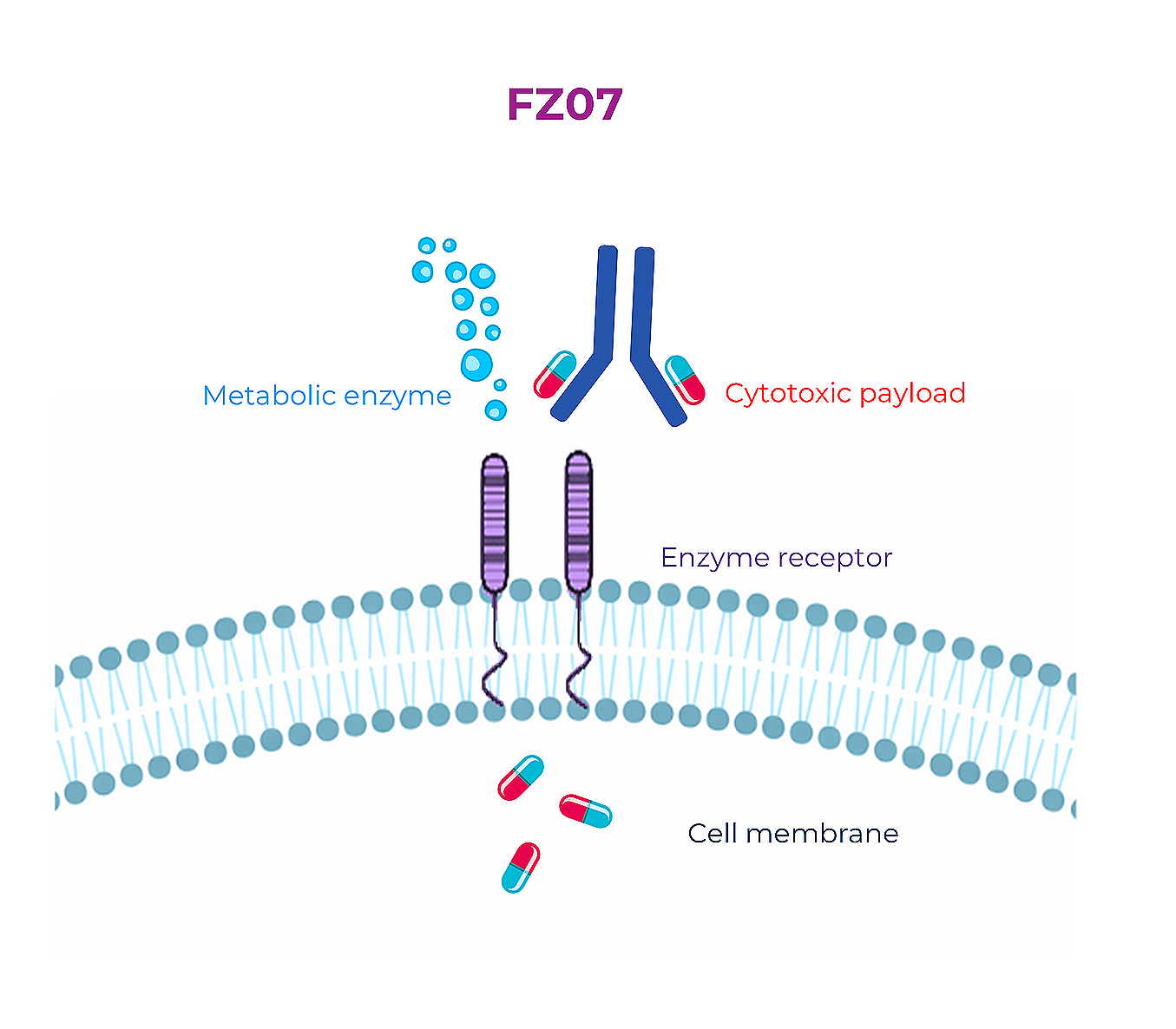
FZ07
FZ07 ADC is a receptor-specific antibody coupled with a cytotoxic payload. The antibody binds to the receptor expressed on the surface of dormant cancer cells. The receptor undergoes endocytosis and delivers the ADC to the lysosome where the payload is released.
FREZENT is interested in delivering a payload that blocks lipid metabolism in the lysosome, bringing a unique biology and first-in-class ADC modality for cancer therapy.
Scientific Publications
Cheng, H., Wang, M., Su, J., Li, Y., Long, J., Chu, J., Wan, X., Cao, Y., & Li, Q. (2022). Lipid metabolism and cancer. Life, 12(6), 784. https://doi.org/10.3390/life12060784
Endo, H., & Inoue, M. (2018). Dormancy in cancer. Cancer Science, 110(2), 474–480. https://doi.org/10.1111/cas.1391
Giancotti, F. G. (2013). Mechanisms governing metastatic dormancy and reactivation. Cell, 155(4), 750–764. https://doi.org/10.1016/j.cell.2013.10.029
Linde, N., Fluegen, G., & Aguirre-Ghiso, J. (2016). The relationship between dormant cancer cells and their microenvironment. Advances in Cancer Research, 132, 45–71. https://doi.org/10.1016/bs.acr.2016.07.002
Recasens, A., & Munoz, L. (2019b). Targeting cancer cell dormancy. Trends in Pharmacological Sciences, 40(2), 128–141. https://doi.org/10.1016/j.tips.2018.12.004
Song, K., Wang, J., & Huang, D. (2023). Therapy-induced senescent tumor cells in cancer relapse. Journal of the National Cancer Center, 3(4), 273–278. https://doi.org/10.1016/j.jncc.2023.09.001
